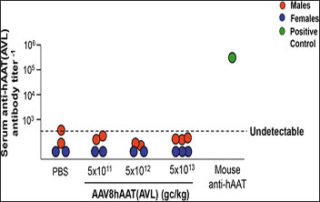
Gene editing of human stem cells to treat rare disease
webmaster2025-10-06T21:52:07+00:00Gene editing has the potential to transform the lives of patients with previously untreatable conditions, offering hope for a better future. As the technology continues to advance, it is likely that we will see more breakthroughs in this field in the coming years, bringing us closer to a world where rare diseases are...